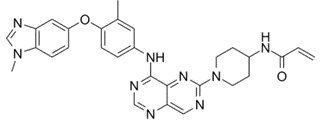
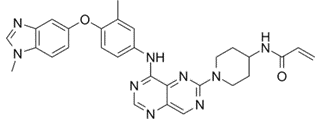
Zongertinib
The FDA has recently granted fast track approval to Zongertinib (Hernexeos®, Boehringer Ingelheim) for adults with unresectable or metastatic non-squamous NSCLC harbouring HER2 (ERBB2) tyrosine kinase domain activating mutations — most commonly the 12 bp exon 20 insertion resulting in YVMA duplication. This approval is particularly impactful for patients who have received prior systemic therapy and developed resistance, providing a crucial new treatment option.
Zongertinib is a novel Tyrosine Kinase Inhibitor (TKI) designed to bind selectively in the ATP binding pocket of HER2. Unlike some earlier pan-ErbB inhibitors that faced limitations due to off-target toxicities (specifically hitting wild-type EGFR), Zongertinib selectively and irreversibly inhibits HER2 without affecting EGFR.
Scientists at Boehringer Ingelheim honed in on a single amino acid difference in the ATP binding pocket between HER2 and EGFR – a Cys residue on HER2 (Cys805). By developing a covalent inhibitor that targets this specific residue, they achieved over 25-fold selectivity for HER2 over EGFR, a critical factor in minimizing side effects and maximizing efficacy. The FDA also approved a companion diagnostic kit, the Oncomine Dx Target test from Life Technologies, to accurately identify these mutations.
A Rapid Path to Patients
The journey of Zongertinib from breakthrough designation to approval was remarkably swift, underscoring the urgency and unmet need it addresses:
August 2024: Breakthrough therapy designation
February 2025: Priority review by FDA
August 2025: Fast track FDA Approval
Expanding Horizons and Future Potential
Beyond NSCLC, Zongertinib's potential continues to unfold. It is currently being investigated for other HER2-mutant cancers, including breast and colorectal cancers, potentially opening up additional therapeutic avenues. Furthermore, combination studies with approved KRasG12C inhibitors sotarasib and adagrasib suggest that Zongertinib may also be effective for cancers that have developed resistance to these drugs, as activation of HER2 and EGFR is associated with KRasG12C inhibitor resistance mechanisms.
A Triumph of Targeted Therapy
Zongertinib's approval marks the third kinase inhibitor drug greenlit in 2025, following the MEK inhibitor Mirdametinib (Gomekli) and the ROS1 inhibitor Taletrectinib (Ibtrozi). This latest milestone reinforces the power of targeted therapy – where a deep understanding of molecular pathways, coupled with innovative drug design approaches like covalent inhibition and leveraging molecular properties, can lead to highly selective, orally administered small molecule drugs that profoundly impact patient outcomes.
https://pmc.ncbi.nlm.nih.gov/articles/PMC11726021/#s2, www.bioprompt.in